February Journal Club
I’m guessing that when most
people think of the Tasmanian Devil, they think of this guy:
That loveable, cuddly (ok, maybe
not so much) character from the Warner Bros. Looney Tunes cartoons.
The cartoon “Taz” had quite the
appetite and left a path of destruction in his wake. His real-life cousins, the
species Sarcophilus harrisii, are not
so destructive or slobbery – and their particular taste for rabbits may not be
clear – but they are carnivorous, speedy, and just about as cute.
 |
| http://upload.wikimedia.org/wikipedia/commons/4/43/Sarcophilus_harrisii_taranna.jpg |
And, unlike the Taz this will be
forever immortalized in cartoon form, the real-life Tasmanian devils are
doomed: Taz’s cousins are becoming extinct because of cancer.
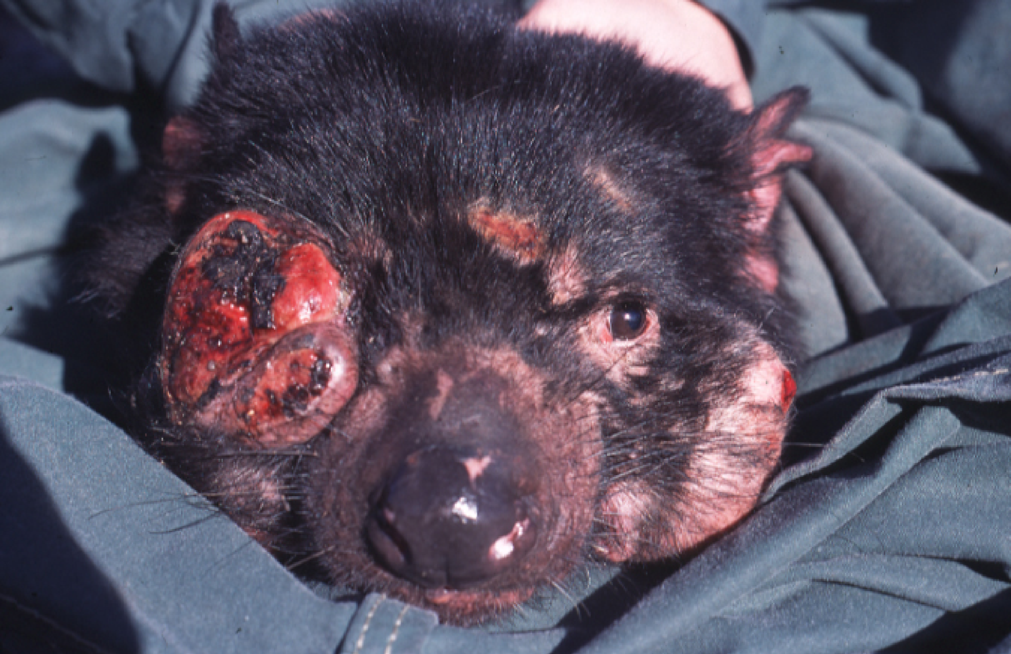
More ferocious than the devil
itself is Tasmanian devil facial tumor disease (DFTD). This cancer causes
disfiguring tumors on the faces of Tasmanian devils and interferes with their
ability to eat, so infected devils actually die of starvation and not from the
cancer directly.
 |
| http://upload.wikimedia.org/wikipedia/commons/9/99/Tasmanian_Devil_Facial_Tumour_Disease.png |
This cancer was first observed in 1996 and has quickly spread
throughout the population of devils on the island of Tasmania, which is part of
Australia. In 2006, a very important observation revealed the
most unique characteristic of DFTD tumors: the tumor cells did not come from host
animal.
Why is this so revealing? In most
cancer types, tumors form because cells of the host somehow go awry. Even if
something from the outside world causes the cancer (UV rays, smoking, human
papillomavirus), the original source of the tumor is from the host, meaning
that the DNA content of the tumor is the same as that of the host. Not exactly
the same – there has to be at least one mutated gene otherwise it would be a
normal cell and not a tumor cell – but close enough that if the DNA sequences
of the host and tumor were compared, it would be clear that the tumor came from
the host.
DFTD is different. In 2006, scientists
found evidence that the DNA sequence from a tumor did NOT match that of its
host, meaning the tumor cells had to come from elsewhere: another animal. And sure
enough, the animals were giving each other cancer through the normal biting
that occurs during mating and feeding. These tumor cells would essentially
graft (stick) themselves onto the face of the victim of the bite and multiply
there. The relative ease with which this occurs explains the rapid spread of
DFTD throughout the Tasmanian devil population.
So, basically, DFTD is the
vampire of the cancer world: it is immortal. Other cancers usually die with
their host. Not DFTD: it lives on in the newly infected animals. Even though
the first devil with the first case of DFTD has long since died (devils only
live 5-6 years), that first tumor has essentially been cloned and started over
with each new infection. As happens over time (think evolution), each clone may
be slightly different than other clones due to random mutations, but all are
variants of the same original tumor. To draw a comparison: when a human cancer
metastasizes, or moves, it stays within the original host and moves from, for
example, the breast to the lymph nodes. DFTD, though, metastasizes from one
animal into another.
This property of being
transmitted from animal to animal has wreaked havoc on the devil population but
can also be used by researchers to study DFTD in the hopes of combating it.
Instead of having to study this tumor in the lab, the devil population acts as
a natural incubator, keeping the tumor cells alive indefinitely. By comparing
the DNA sequences of normal devils to the sequences of present-day DFTD tumors,
the following key questions can be studied:
1.
What makes the tumor cells different than normal
devil cells and how is it able to cause disease?
2.
How has the cancer changed over time from the
initial founder to the different lineages now found in nature?
3.
How did the cancer spread from one devil to the
entire devil population?
These are precisely the questions
raised by a group of researchers when they set out to sequence the genome of
the Tasmanian devil and different DFTD tumors (Murchison EP, et al. (2012). Genome Sequencing and Analysis of the Tasmanian Devil and Its Transmissible Cancer. Cell 148(4): 780-791).
How can sequencing the genome of
an animal and its cancer cells reveal so much about the history of the disease
and how it works? It all has to do with comparing different sequences and
analyzing what must have happened over the course of time to cause these
changes.
Three genomes were sequenced: a
normal, adult female devil and two tumors from different parts of Tanzania (in
the paper, these are called the titillating names of 87T and 53T). The two
tumors have undergone random mutations since they diverged from the original
DFTD tumor. By comparing these mutations to each other, researchers can
estimate what changes occurred after
the tumors left the original host. If both 87T and 53T have the same mutation
in gene X, this mutation probably was in the original tumor because it is not
statistically likely that both tumors would separately have the exact same
mutation in the exact same place. (Consider these odds: if the tumor DNA genome
is about 3 billion units in size, then the chance of an identical mutation in 2
different genomes is a 1/3,000,000,000 x 1/3,000,000,000 = 1 in 9,000,000,000,000,000,000
chance. Take those odds to Vegas!)
By identifying the mutations
present in the tumors and not in the normal devil genome, the original tumor
DNA sequence could be deduced. Then, by comparing the tumor genome to the
normal devil genome, the differences that make the normal cells “normal” and
the tumor cells “tumorigenic” were revealed. For example, mutations were found
in genes that are related to cancer in humans. Also, mutations were found in
genes that would allow the tumor to “hide” from the host immune system, a
property it has to have to be able to survive on the face of a devil.
Once the researchers identified
where the differences were in the sequences of the two tumor genomes, these
“hotspots” could be identified in hundreds of tumors throughout Tanzania. A
sort of family tree was constructed to trace the spread of DFTD from the
original devil to its present widespread distribution – a history of the
disease.
Understanding the past
development of DFTD is academically interesting, as is knowing what the
original tumor “looked” like, but there are further implications beyond just
basic curiosity. By knowing how DFTD evolved in the past, scientists can
predict how it may change in the future and act to target these changes to
prevent or stop the disease. Furthermore, there are still ways in which the
data from this study can be analyzed. For instance, even though mutations were
found in certain genes, the “how” question has not been answered: how these
mutations cause DFTD. So far, only genes that are known to play a role in human
cancers have been probed for the presence of mutations, but others could
possibly be involved.
DFTD is well on its way to
causing the extinction of an entire species. Understanding how the tumor cells
are different than normal cells is key for developing therapy to cure or
prevent DFTD. One other cancer is caused in the same way as DFTD (that is, is
immortally transmitted through bites): a cancer of dogs, called canine
transmissible venereal tumor (CTVT). The lessons learned from DFTD may be
relevant to CTVT. So, if the soft spot in your heart for Taz isn’t big enough,
think of Fido.
No comments:
Post a Comment